- The major toxic air pollutants that Environmental Monitoring Professionals assess are sulfur dioxide, nitrogen dioxide, carbon monoxide, ozone, particulate matter, and lead.
- Other serious toxic gases also monitored are ammonia, VOCs, hydrogen sulfide, and hydrogen chloride.
- The concentrations of toxic air pollutants are regulated by strict standards to limit health problems and environmental damage.
The World Health Organization (WHO) ranks air pollution as the second-highest public health risk, contributing to noncommunicable diseases and the number-one environmental risk for child health. Outdoor pollution affects all countries, high-, middle-, and low-income. Environmental monitoring can help assess pollution levels and support policy development and implementation to reduce public health risks. This article discusses the major toxic gases that Environmental Monitoring Professionals track to reduce human health risks from outdoor air pollution.
What is Environmental Monitoring
Environmental monitoring is necessary to protect public health, the environment, cultural monuments, and infrastructure. Environmental risks threaten humans, ecosystems, and manmade structures.
Environmental monitoring involves continuously tracking environmental changes and encompasses all procedures and actions that use systems, methods, and models developed for this purpose. Environmental monitoring serves many purposes at national and international levels for policy development and implementation, and at industry levels for regulation.
- Policymaking: Easily accessible data can be integrated into assessment and reporting routines and information management systems to enable informed decision-making at national and international levels. Sensors can provide data on specific pollutants and multiple pollutants, and identify the sectors responsible for pollution. Public authorities use the information to guide national policy, set and revise emission standards, and regulate polluting activities.
- Compliance monitoring: Industrial facilities are under pressure to monitor and report their emissions to reduce their environmental footprint and comply with local, national, and international regulatory standards.
In the USA, the Environmental Protection Agency (EPA) monitors and regulates chemicals that pollute the air, water, and soil.
Air Pollutants Assessed in Environmental Monitoring
Air pollutants are monitored for their toxicity, corrosivity, asphyxiating properties, and reactivity. Most air pollutants are colorless and odorless, making them difficult to detect and smell, and must be monitored with specially designed detectors capable of measuring ppm (parts per million) and ppb (parts per billion) levels at which they become harmful. On-site monitoring and control of toxins for environmental assessment, a necessary process, were challenging and constrained by conventional laboratory-based techniques. New portable air sensors that accurately assess pollutants in real time are now providing valuable alternatives to laboratory-based techniques for environmental monitoring.
Air pollution, both indoor and outdoor, has serious health impacts. Air pollutants can aggravate asthma and respiratory infections in children. Among adults, air pollution causes high rates of respiratory and cardiovascular diseases and cancers, increasing morbidity and mortality.
Outdoor Air Pollutants
Sources of outdoor air pollutants can be anthropogenic or natural, as shown in Figure 1.
- Natural emissions: These are produced by Earth’s crust, such as radon gas from radioactive decay and volcanic activity. Hydrogen sulfide from the decomposition of organic material in wetlands is another example.
- Anthropogenic emissions: Emissions from human activity can come from stationary or mobile sources. Industrial factories, mines, houses, thermal power stations, forest and agriculture fires, and the use of paints, varnish, etc., are stationary sources. Emissions from vehicles, trains, ships, and airplanes are the main mobile sources.
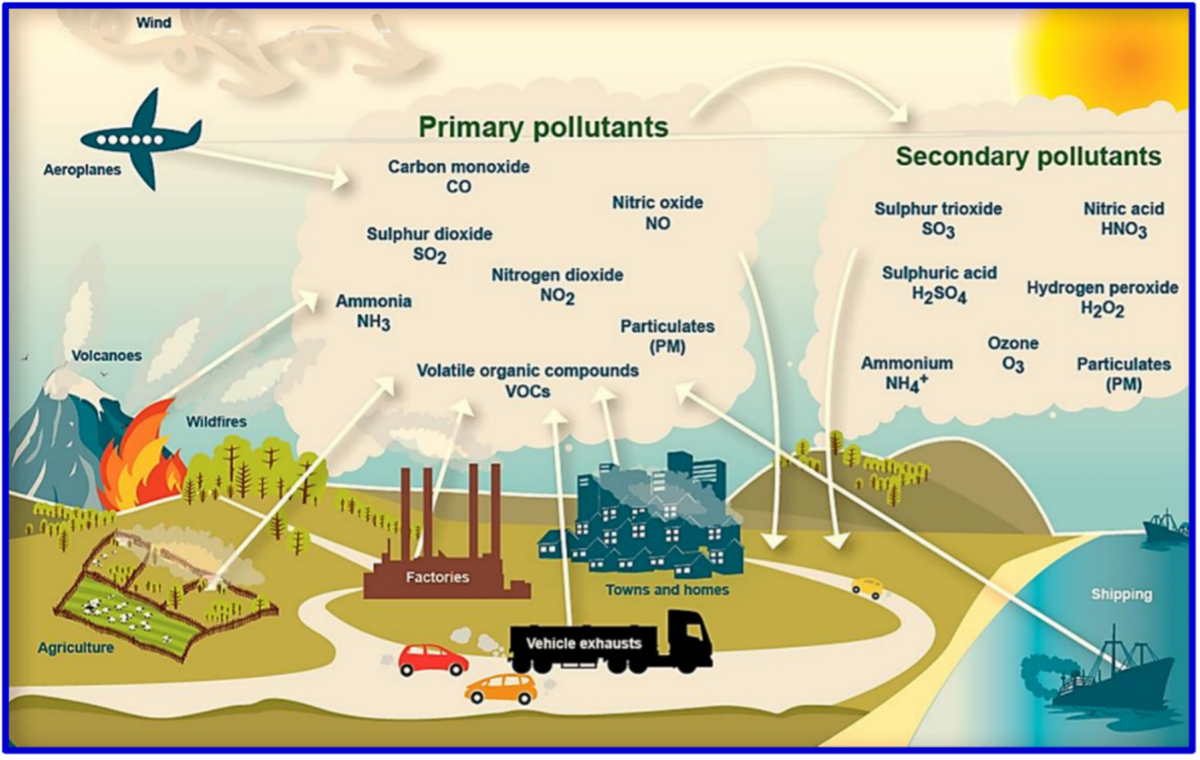
Figure 1: The major gaseous pollutants that environmental professionals monitor, Dhall et al. (2021). (Image credits: https://doi.org/10.1016/j.sintl.2021.100116)
A number of hazardous outdoor air pollutants are identified under the Clean Air Act. In the USA, under the Clean Air Act, the Environmental Protection Agency (EPA) has to formulate national ambient air quality standards (NAAQS) for six pollutants considered harmful: sulfur dioxide, nitrogen dioxide, carbon monoxide, ozone, particulate matter, and lead. Two types of standards exist for these six principal or criteria air pollutants: primary and secondary limits, which should not be exceeded. Gas concentrations above these levels are harmful to people and the environment. Table 1 gives details of these standards.
- Primary standards: These standards provide public health protection, especially for vulnerable populations such as the elderly, children, and people with asthma.
- Secondary standards: These standards protect crops, vegetation, animals, and buildings.
These levels are regulated by the National Emissions Standards for Hazardous Air Pollutants program.
Table 1: “National Ambient Air Quality Standards (40 CFR part 50) for six principal pollutants (“criteria” air pollutants),” EPA. (Credits: https://www.epa.gov/criteria-air-pollutants/naaqs-table)
| Pollutant [links to historical tables of NAAQS reviews] |
Primary/ Secondary |
Averaging Time | Level | Form | |
| Carbon Monoxide (CO) | primary | 8 hours | 9 ppm | Not to be exceeded more than once per year | |
| 1 hour | 35 ppm | ||||
| Lead (Pb) | primary and secondary (1) |
Rolling 3-month average | 0.15 μg/m3 | maximum arithmetic mean of 3 consecutive monthly means in a 3-year period | |
| Nitrogen Dioxide (NO2) | primary | 1 hour | 100 ppb | Annual 98th percentile of 1-hour daily maximum concentrations, averaged over 3 years | |
| primary and secondary |
1 year | 53 ppb (2) | Annual Mean | ||
| Ozone (O3) | primary and secondary (3) |
8 hours | 0.070 ppm | Annual fourth-highest daily maximum 8-hour concentration, averaged over 3 years | |
| Particle Pollution (PM) | PM2.5 | primary | 1 year | 9.0 μg/m3 | annual mean, averaged over 3 years |
| secondary | 1 year | 15.0 μg/m3 | annual mean, averaged over 3 years | ||
| primary and secondary |
24 hours | 35 μg/m3 | 98th percentile, averaged over 3 years | ||
| PM10 | primary and secondary |
24 hours | 150 μg/m3 | Not to be exceeded more than once per year on average over 3 years | |
| Sulfur Dioxide (SO2) | primary (4) | 1 hour | 75 ppb | Annual 99th percentile of 1-hour daily maximum concentrations, averaged over 3 years | |
| secondary (5) | 1 year | 10 ppb | annual mean, averaged over 3 years | ||
In addition to these six chemicals, environmental monitoring professionals also check for volatile organic compounds (VOCs), hydrogen chloride, hydrogen sulfide, and ammonia. See Figure 1 for the list of main air pollutants.
-
Sulfur Dioxide
EPA standards aim to prevent harmful exposure to all sulfur oxides (SOx). However, sulfur dioxide (SO2), a colorless and odorless gas, is the cause of greatest concern. It is produced by burning fossil fuels in power plants, industrial facilities, and mining and extraction operations, and to a lesser extent by volcanoes. Mobile sources burn fuels with high sulfur content, releasing sulfurous gases. High concentrations of SO2 in the air lead to the formation of other sulfur oxides. SO2 also combines with compounds to form minute particles that contribute to particulate matter (PM) pollution. The deleterious effects of SO2 are as follows:
Health effects: SO2 can irritate skin, eyes, and the respiratory tract. High concentrations of SO2, especially during heavy physical activity, cause inflammation of the respiratory system, leading to breathing problems, coughing, and lung dysfunction, and can worsen pre-existing heart and respiratory conditions. Asthmatics and children are at greater risk from the gas and PM it creates.
Environmental effects: High concentrations of SOx can damage plant and tree foliage and growth. SO2 and other SOx form acid rain when they dissolve in rain, causing widespread damage to natural ecosystems, national parks, wilderness areas, stone, and monuments. PM formed from SO2 can lead to haze that reduces visibility.
-
Nitrogen Dioxide
Nitrogen oxides (NOx) are very reactive gases, and NO2 is one of them. The others are nitrous oxide (N2O) and nitric oxide (NO). NO2 is a reddish-brown gas with a pungent odor, formed from burning fossil fuels in power plants, industrial facilities, and mobile sources. NO2 absorbs solar radiation, reducing air transparency and warming the air. It is very reactive and generates oxidizing gases in the atmosphere. All nitrogen oxides react with other compounds to form secondary pollutants, such as particulate matter and ozone. The health and environmental impacts of NO2 are listed below:
Health effects: Even short-term exposures of minutes to months can affect the respiratory system, aggravating asthma and other respiratory diseases and leading to hospital visits. Long-term exposure to high NO2 levels causes asthma and respiratory infections. The elderly and children are at greater risk from NO2 pollution. NO2 is used as a proxy for traffic-related pollutants and contributes to mortality from respiratory and cardiovascular causes.
Environmental effects: NO2 and other nitrogen oxides react with oxygen, water, and other chemicals to form acid rains, that harms ecosystems and lakes. In the coastal waters, it causes nutrient pollution. NOx in PM causes haze and reduces visibility.
-
Carbon Monoxide
Carbon monoxide (CO) is released from the incomplete combustion of fuels. The largest contributor to CO pollution is the use of fossil-fuel vehicles, trucks, and machinery. It is also emitted by power and energy-generating plants. Stoves, furnaces, and room heaters in homes and buildings can also emit CO and deteriorate indoor air quality. CO becomes a pollutant outdoors in busy urban traffic zones and spaces with little ventilation. CO is colorless and odorless.
Health effects: High concentrations of CO reduce oxygen delivery to organs in humans because hemoglobin in the blood has a 300-fold greater affinity for CO than for oxygen. Exposure to low concentrations leads to headache, nausea, and dizziness, and at very high concentrations can cause drowsiness, chest pain, pulmonary edema, brain changes, and death.
Environmental effects: CO is involved in reactions that form ground-level ozone, a greenhouse gas. CO in its own right also affects the climate, so limiting it is considered a climate change mitigation measure.
-
Ground-level Ozone

Figure 2: “Formation of ozone,” EPA. (Image credits: https://www.epa.gov/ground-level-ozone-pollution/ground-level-ozone-basics#wwh)
While stratospheric ozone is beneficial, (tropospheric or) ground-level ozone is harmful because it has adverse health effects on people. Ozone is not released directly into the air, but is created during reactions among SO2, NO2, CO, and VOCs in the presence of sunlight and heat, as shown in Figure 2. These primary pollutants are released by vehicles, power plants, and industries that use boilers, refineries, and chemical facilities. Ozone (O3) is formed year-round and can reach unhealthy levels, especially on sunny, warm days in urban centers. From cities, ozone is transported long distances to reach rural areas and natural ecosystems. Ozone health and environmental effects are as follows:
Health effects: The ozone effects depend on exposure duration and concentration. Effects can start with coughing and a sore throat and progress to difficulty in breathing, pain, and damage to the airways. Lungs become more susceptible to infection, and existing lung diseases and asthma become more frequent.
Environmental effects: Exposure to high levels of ozone affects vegetation in forests, national parks, and wildlife refuges. Ozone can be particularly harmful to plant growth.
-
Particulate Matter
Particle pollution or particulate matter (PM) is a mixture of solid particles and liquid droplets. Particles vary in size and shape and are composed of hundreds of different chemicals. Some particles are formed directly and released from construction sites, farms, fires, or industrial stacks. Most particles result from chemical reactions among primary pollutants, such as SO2 and NO2, and other compounds. PM consists of sulphates, nitrates, mineral dust, ammonia, black carbon, and sodium chloride.
Smoke, dust, and soot are examples of PM that humans can see. However, PM can be very small, which can only be seen with microscopes, and it is a major pollutant. There are two types of small PM based on size:
- 5: These are very fine particles with a diameter of 2.5 micrometers or less.
- PM10: These particles are below 10 micrometers and also include PM2.5.
The health and environmental impacts of PM are as follows:
Health effects: Both PM 2.5 and PM10 can be inhaled and cause severe health problems. Because it is heavy, PM10 settles in the airways. However, PM2.5 is carried into the lungs, enters the bloodstream, and reaches other organs. PM2.5 is linked to respiratory and cardiovascular problems, lung cancer, pneumonia, strokes, cerebrovascular disease, hypertension, diabetes, dementia, and chronic kidney disease. PM2.5 accounts for the largest share of negative health effects from air pollution. Children and infants are at the greatest risk because they inhale more air per unit of body weight and spend more time outdoors.
Environmental effects: Particulate matter causes haze and reduces visibility. It adversely affects ecosystems because it is deposited in soil and water and taken up by plants, where the various chemicals that form PM can harm plant growth and yield. PM affects the climate: some components increase air warming, while others can cool it, depending on their composition.
-
Lead
Lead (Pb) emissions sources can vary, with lead smelters producing the highest levels. Other major sources are ore and metal processing and the use of leaded aviation fuel in aircraft. Minor sources are lead-acid battery manufacturers, waste incinerators, and utilities. Lead health and environmental effects are as follows:
Health effects: After inhalation, lead is distributed through the bloodstream and accumulates in the bones. Lead has adverse effects on the kidneys, nervous system, immune system, reproductive system, developmental system, and cardiac system. Lead also affects the blood’s oxygen-carrying capacity. Children and infants are very sensitive to lead exposure and develop behavioral problems and learning deficits.
Environmental effects: Lead persists in the environment and can be deposited in soils. High lead levels in the air can also have neurological effects in vertebrates and affect growth and reproduction in plants and animals.
-
Volatile Organic Compounds
Volatile organic compounds (VOCs) are pollutants found both indoors and outdoors. VOCs such as benzene, xylene, and toluene, which are used in many products, are also found outdoors. VOCs, which are liquids or solids, vaporize at ambient temperatures and pressures and are released during manufacturing or daily use.
Earlier, the EPA categorized outdoor organic compounds as “Reactive Organic Gases” (ROG), but they have now changed the term to VOC. The reasons for regulating VOCs outdoors differ from those for regulating VOCs indoors and mainly concern their role in forming photochemical smog.
Environmental effects: In the USA, ambient VOC levels are regulated because they combine with other primary pollutants, such as NOx and CO, in the presence of sunlight to form smog. Only reactive VOCs need to be monitored to comply with EPA rules. VOCs can also spill and contaminate soils, enter drinking water, and emit vapors that can enter buildings, worsening indoor air quality.
Health effects: Smog affects the eyes, nose, and throat and can worsen pre-existing respiratory and cardiac conditions. Elderly people, children, people with heart or lung problems, and those exercising or spending time outdoors are at greater risk from smog. Smog also disrupts tourism due to low visibility. Exposure to some VOCs can affect the eyes, nose, throat, liver, kidneys, and the central nervous system, and some organic compounds can cause cancer.
-
Ammonia
Ammonia (NH3) is a major air pollutant because it contributes to the formation of particulate matter. The main source is agriculture, which accounts for nearly 80% of ammonia pollution in Europe. There is four times as much ammonia in rural areas as in cities, and it comes from nitrogen fertilizers and animal waste. The next source is fossil fuel exhaust from vehicles, followed by emissions from industry, waste, and combustion processes. Ammonia produced on the farms can move to nearby urban areas. Increasing ambient temperature increases ammonia pollution in urban areas. NH3 is a toxic gas with a pungent smell.
Health effects: Ammonia can cause asthma in children in rural areas, especially among those living near intensive pig farms. However, the main health impact comes from the particulate matter. Ammonia accounts for 47% of particulate matter, and all the resulting adverse health effects of PM, including premature deaths.
Environmental effects: Ammonia gas can be deposited on vegetation in high concentrations. It affects wild plants in meadows and forests, leading to the loss of sensitive species such as lichens, liverworts, and mosses, and reducing biodiversity. It also increases the incidence of pests and diseases. Ammonia gas can contaminate water bodies and cause eutrophication, which promotes toxic algal blooms that deplete oxygen and lead to fish kills. It also causes soil acidity in the natural environment.
-
Hydrogen Chloride
Hydrochloric acid (HCl) is classified as a “federal hazardous air pollutant.” It is released during the burning of fuels and waste, metal scrap smelting, dehalogenation processes, and the pyrolysis of several wire insulation materials, such as chlorinated acrylics, polyvinyl chloride, and retardant-treated compounds. The industries producing HCl pollutants are electric services, inorganic chemicals, guided-missile, and space-vehicle production. Diffuse emission from the use of HCl for pH adjustment in swimming pools and spas, and as disinfectant in bathrooms, kitchens, hospitals, households, and commercial establishments, also occurs. HCl effects are as follows:
Health effects: Hydrochloric acid is extremely corrosive and irritates the eyes and the respiratory tract. Acute exposure causes inflammation and ulceration of the respiratory system, coughing, chest pain, and pulmonary edema. Long-term exposure to even small amounts of acid causes dental erosion.
Environmental effects: HCl emissions are carried long distances by wind and deposited on soil, contaminating groundwater. It is also incorporated into fog and rain, producing acid rain. It is involved in processes that form smog. HCl is highly toxic to all life forms. It reacts with alkaline materials, dissolving limestone, clay, and igneous rocks, causing damage to structures, buildings, bridges, and art made of these materials.
-
Hydrogen Sulfide
Hydrogen sulfide (H2S) from natural sources (volcanoes, wetlands, & estuaries), along with SO2, shapes Earth’s climate. Anthropogenic sources are increasing and come from pulp and paper industries, gas refineries, landfills, wastewater treatment plants, sewage systems, and geothermal power plants, affecting people living near them. H2S pollution health and environmental effects are as follows:
Health effects: H2S is regulated primarily because of its odor nuisance. However, it does also have health effects. Acute and short exposures of 30 minutes to 2 ppm can affect asthmatics. Around low concentrations (20-50 ppm), H2S irritates the eyes and upper respiratory tract, and prolonged exposure can cause pulmonary edema. At higher concentrations (200-300 ppm), it is life-threatening. H2S is heavier than air and settles near the ground, so children are at higher risk of exposure to H2S pollution.
Environmental effects: In the air, H2S reacts to form SO2, sulfuric acid, and particulate matter. It remains in the atmosphere for 18 hours. H2S can diffuse in water, to which fish and other freshwater animals are more sensitive than marine animals. Plants’ growth and survival can be negatively affected by H2S, but in some cases, it can serve as a source of sulfur.
In addition to these gases, carbon dioxide is continuously monitored because it causes climate change and all its associated negative impacts on the environment, biodiversity, food production, and human life. Although it is a confirmed indoor toxic health hazard, it is not reported as a direct health hazard in outdoor pollution; however, it can worsen many existing health conditions.
Sensors for Measuring Outdoor Pollutants
Industries that emit pollutants directly or that could produce secondary pollutants should monitor emissions at the stack and gate to ensure they do not pollute the environment. Industries can use fixed devices for continuous monitoring, while Environment Monitoring Professionals rely on portable devices for environmental assessment, industrial emissions monitoring, and pollution control. Interscan produces fixed and portable tools for precise, real-time measurements of over 20 gases, including most air pollutants, with data-logging capability. The company supplies both fixed and portable devices for use in industries and environmental monitoring to keep people and ecosystems safe.
Schedule a meeting for a demo of Interscan’s fixed and portable gas devices for environmental assessment of air pollutants.
Sources
ARB.CA.Gov. (1997). Hydrochloric acid. Retrieved from https://ww2.arb.ca.gov/sites/default/files/classic/toxics/tac/factshts1997/hydchlor.pdf
ARB.CA. Gov. (n.d.). Hydrogen Sulfide & Health. Retrieved from https://ww2.arb.ca.gov/resources/hydrogen-sulfide-and-health
ARB.CA.Gov. (n.d.). Inhalable Particulate Matter and Health (PM2.5 and PM10). Retrieved from https://ww2.arb.ca.gov/resources/inhalable-particulate-matter-and-health
Ausma, T., & De Kok, L. J. (2019). Atmospheric H2S: impact on plant functioning. Frontiers in Plant Science, 10, 743.
Ausma, T., & De Kok, L. J. (2019). Atmospheric H2S: Impact on Plant Functioning. Frontiers in plant science, 10, 743. https://doi.org/10.3389/fpls.2019.00743
Călămar, A. N., Găman, G. A., Pupăzan, D., Toth, L., & Kovacs, I. (2017). Analysis of environmental components by monitoring gas concentrations in the environment. Environmental Engineering & Management Journal (EEMJ), 16(6).
DCCEEW.Gov.Au. (2022, June 30). Substances. Retrieved from https://www.dcceew.gov.au/environment/protection/npi/substances/fact-sheets/hydrochloric-acid
Dhall, S., Mehta, B. R., Tyagi, A. K., & Sood, K. (2021). A review on environmental gas sensors: Materials and technologies. Sensors International, 2, 100116.
EPA. (2025, March 11). Ground-level Ozone Basics. Retrieved from https://www.epa.gov/ground-level-ozone-pollution/ground-level-ozone-basics#wwh
EPA. (2025, May 30). What is PM, and how does it get into the air? Retrieved from https://www.epa.gov/pm-pollution/particulate-matter-pm-basics#PM
EPA. (2025, June 17). Basic Information about Carbon Monoxide (CO) Outdoor Air Pollution. Retrieved from https://www.epa.gov/co-pollution/basic-information-about-carbon-monoxide-co-outdoor-air-pollution#What%20is%20CO
EPA. (2025, July 10). Basic Information about NO2. Retrieved from https://www.epa.gov/no2-pollution/basic-information-about-no2#What%20is%20NO2
EPA. (2025, July 18). Basic Information about Lead Air Pollution. Retrieved from https://www.epa.gov/lead-air-pollution/basic-information-about-lead-air-pollution
EPA. (2025, Sept 8). Technical Overview of Volatile Organic Compounds. Retrieved from https://www.epa.gov/indoor-air-quality-iaq/technical-overview-volatile-organic-compounds
EPA. (2026, Jan 6). Sulfur Dioxide Basics. Retrieved from https://www.epa.gov/so2-pollution/sulfur-dioxide-basics#what%20is%20so2
EPD. (2025, May 26). Volatile Organic Compounds and Smog. Retrieved from https://www.epd.gov.hk/epd/english/environmentinhk/air/prob_solutions/vocs_smog.html
Ho, C. K., Robinson, A., Miller, D. R., & Davis, M. J. (2005). Overview of Sensors and Needs for Environmental Monitoring. Sensors, 5(1), 4-37. https://doi.org/10.3390/s5010004
Kumar, A., Kim, H., & Hancke, G. P. (2012). Environmental monitoring systems: A review. IEEE Sensors Journal, 13(4), 1329-1339.
Krzyzanowski, M. (2023). The health impacts of nitrogen dioxide (NO2) pollution. Retrieved from https://www.env-health.org/wp-content/uploads/2023/06/NO2_briefing_EN.pdf
Liu, S., Xu, H., Wang, J., Ding, J., Liu, P., Yang, Y., & Liu, L. (2024). Evidence for global increases in urban ammonia pollution and their drivers. Science of the Total Environment, 955, 176846.
Minnesota Pollution Control Agency. (n.d.). Air pollutant Volatile organic compounds (VOCs). Retrieved from https://www.pca.state.mn.us/pollutants-and-contaminants/volatile-organic-compounds-vocs
Okobia, L. E., Hassan, S. M., & Peter, A. (2017). Increase in outdoor carbon dioxide and its effects on the environment and human health in Kuje FCT Nigeria. Environmental Health Review, 60(4), 104-112.
Sangkham, S., Phairuang, W., Sherchan, S. P., Pansakun, N., Munkong, N., Sarndhong, K., … & Sakunkoo, P. (2024). An update on adverse health effects from exposure to PM2. 5. Environmental Advances, 18, 100603.
Saxena, P., & Shukla, P. (2023). A review on recent developments and advances in environmental gas sensors to monitor toxic gas pollutants. Environmental Progress & Sustainable Energy, 42(5), e14126.
Taha, S. S., Idoudi, S., Alhamdan, N., Ibrahim, R. H., Surkatti, R., Amhamed, A., & Alrebei, O. F. (2025). Comprehensive Review of Health Impacts of the Exposure to Nitrogen Oxides (NOx), Carbon Dioxide (CO2), and Particulate Matter (PM). Journal of Hazardous Materials Advances, 100771.
UCAR. (n.d.). Volatile Organic Compounds (VOCs). Retrieved from https://scied.ucar.edu/learning-zone/air-quality/volatile-organic-compounds#:~:text=Gasoline%20and%20natural%20gas%20are,ozone%2C%20another%20harmful%20air%20pollutant.
UNECE. (n.d.). Environmental Monitoring. Retrieved from https://unece.org/environmental-monitoring#


