- Chlorine should be monitored in industries that use it as a raw material for manufacturing, as a disinfectant, and as a bleaching agent.
- Chlorine should be monitored because it is toxic, corrosive, and a fire risk.
- Chlorine monitors should be placed in places where the chemical is transported, stored, fed into equipment, and processed.
Among occupational hazardous gases, chlorine causes the third-highest number of injuries and deaths in industry. Chlorine is a crucial industrial agent and raw material, but it is an occupational hazard that should be handled with care. Most chlorine accidents occurred in industries producing paper, leather, lumber, chemicals, and petroleum. However, chlorine must be monitored in several other industries. This article lists the industries where chlorine emissions are commonly found.
Why Chlorine Gas is an Occupational Hazard
Chlorine (Cl2) is an occupational hazard because it is toxic and a strong oxidizer. Its toxicity causes health problems; its oxidizing properties pose fire and corrosion risks.
- Health risks: Chlorine becomes a health hazard through contact and inhalation. When the gas comes in contact with moist tissue, it forms hydrochloric acid. Acute exposure to low levels can cause eye irritation, salivation, and sneezing; high concentrations can cause permanent eye damage, chest pain, pulmonary edema, pneumonia, asphyxiation, and death. Evaporation of liquid Cl2 causes frostbite. It also leads to first-degree burns and, through long exposures, to secondary burns. Repeated chronic exposure to Cl2 can cause tooth erosion, a skin rash, and permanent lung damage.
- Fire risks: Cl2 gas is nonflammable, but because it is a strong oxidizer, combustibles and /or organics such as paper, wood, and oil can catch fire on contact with Cl2 and produce toxic chemicals. Chlorine should be handled with care, stored away from sunlight and added remotely when possible, and protected from potential vehicle impact. It should be kept away from flames or chemicals with which it reacts, such as ammonia, hydrocarbons, etc.
- Corrosion risks: Chlorine is corrosive because it reacts easily with iron, tin, copper, and silver alloys, even at low concentrations. At higher concentrations, it oxidizes the metal. So, pipes and equipment used to store, transport, and handle the gas and liquid can be weakened, damaged, develop leaks, malfunction, or cause accidents. The high pressure at which it is stored can increase the risk of an explosion.
Chlorine can be used as a gas or a liquid in industries. It is a gas at ambient temperature and pressure and can be liquified by cooling. The greenish-yellow gas has a bleach-like odor and is heavier than air, causing it to sink to the bottom. The amber-colored Cl2 liquid is heavier than water. When added to water, it boils and sinks to the bottom. Chlorine is usually transported and stored as a liquid under pressure in containers. When liquid Cl2 leaks at ambient temperature and pressure, it turns into gas in a 1:450 ratio, i.e., 1 liter of chlorine liquid yields 450 liters of Cl2 gas. Hence, industries that use liquid chlorine must also monitor the air for gaseous chlorine in the event of an accidental release.
When liquid Cl2 leaks, it should not be doused with water, as it can react with water to form hydrochloric acid. Water can be sprayed on Cl2 gas to contain it.
Why Chlorine Should be Monitored in Industries
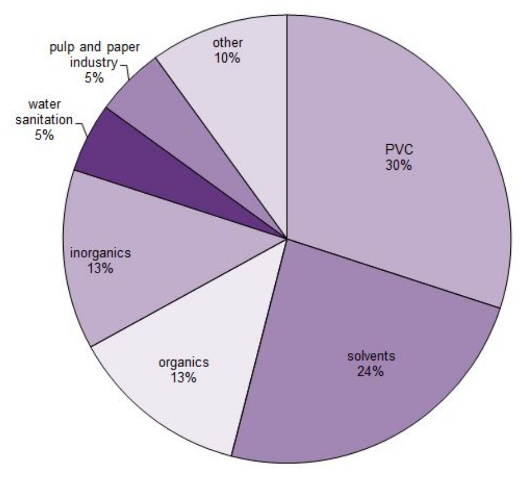
Figure 1: “Uses for chlorine in 2018,” Essential Chemistry Industry.Org. (Image credits: https://www.essentialchemicalindustry.org/chemicals/chlorine.html)
Chlorine is used as a raw material for manufacturing, as a bleaching agent, and as a disinfectant; see Figure 1. Chlorine is the most commonly used disinfectant for water. The chlorine market was valued at USD 39.95 billion in 2025, and demand is expected to grow at a CAGR of 4.5% till 2035.
Industries should monitor chlorine to protect their workers, comply with safety standards, and improve process efficiency.
- Health and safety risk: Employees can be exposed to chlorine during transport, handling, and accidental release. An accidental release can affect people in the industry and the surrounding communities.
- Compliance: Industries that produce, use, or transport chlorine must comply with strict safety standards. The Occupational Safety and Health Administration (OSHA) stipulates that exposure levels should not exceed 1 ppm at any time.
- Industrial efficiency: Monitoring chlorine concentration can also improve disinfection and chemical production efficiency.
The 12 common industries that use Cl2 gas and should monitor the gas are discussed below.
-
Chemical Manufacturing and Processing
Chlorine is a raw material essential to the production of many chemicals, solvents, polymers, agrochemicals, and drugs.
- Polymers: Nearly 30% are used to produce Polyvinyl Chloride (PVC). Other polymers produced from chlorine include poly(chloroethene) and intermediates, such as isocyanates, used in polyurethanes.
- Solvents: Around 24% of chlorine is used to make solvents such as trichloroethene, which is used in dry cleaning.
- Inorganic chemicals: About 13% of chlorine is needed to produce inorganic chemicals such as hydrogen chloride, hydrochloric acid, titanium dioxide, and chlorinated compounds.
- Organic chemicals: Some organic chemicals made from the 13% of chlorine are oxygenates, such as epoxypropane (propylene oxide), propane-1,3-diol to produce polyols, 1-Chloro-2,3-epoxypropane, and chloromethanes. The latter, in turn, is used to produce silicone and poly(tetrafluoroethene).
Chlorine is also needed to produce agrochemicals such as pesticides, herbicides, insecticides, and fungicides.
The production of chlorine and other chlorine-derived chemicals requires careful monitoring of the gas in storage, processing, and handling areas for safety, and during production to ensure accurate and efficient operation.
-
Pharmaceuticals
Chlorine is widely used in pharmaceutical products. Around 25% of pharmaceutical products contain chlorine. Over 60% use chlorine in the manufacturing process because the production relies on smaller chlorine-containing intermediates that lose their identity as they form larger chemical molecules.
So, chlorine chemistry is involved in the production of 88% of top-selling drugs of all therapeutic classes. These can be prescription and over-the-counter drugs, including painkillers, antihistamines, antibiotics, and decongestants.
Chlorine gas monitoring is needed in the storage, feeding, and processing areas to keep workers safe.
-
Municipal Drinking Water and Wastewater Treatment
Chlorine treatment for drinking water is a standard procedure despite its risks, because it is low-cost. Chlorine is a disinfectant that kills many waterborne pathogens and algae. Moreover, its strong oxidizing powers remove many organic and inorganic compounds that pollute water. It can remove offensive smells and improve the taste of water. Chlorination gives reliable results and provides flexibility in dosage. However, because some microbes are developing resistance to chlorine, the gas dosage is increasing.
In wastewater treatment plants, chlorine is added as a gas or liquid bleach (sodium hypochlorite) to kill harmful pathogens (bacteria and viruses) in sewage and industrial effluents, making it safe for discharge into the environment. Chlorine is also useful in oxidizing organic and inorganic matter, including agricultural waste and pharmaceutical compounds. However, Cl2 toxicity and corrosiveness are a problem. Chlorine use results in iron deposition in water. Residual chlorine left in water can be toxic to people and fauna. Chlorine gas detectors are necessary near its containers, dosing pump, mixer, and sampling area to protect workers.
The increasing adoption of water disinfection in developing countries is a major factor driving demand for chlorine, according to 2026 market research by EMR.
-
Aquatic Industry
Water quality is crucial for aquaculture operations, particularly for sanitation and biofilm removal. Chlorine is used to eradicate harmful microbes in the source water. Depending on the operations, the areas treated with chlorine to remove pathogens may differ. These can be hatcheries, tanks, pipelines, feeding systems with algae and artemia, and nets. All equipment, surfaces, screens, and containers are disinfected. Before aquaculture water is released into natural environments, it is treated with chlorine again. Therefore, chlorine must be monitored at various stages to maintain fish health and minimize environmental impacts.
Monitoring chlorine levels is essential to ensure that the amounts supplied are sufficient to eradicate harmful organisms without being so high as to harm fish or other aquatic organisms being grown, as chlorine is deleterious to several fish species. According to the FAO, free chlorine is lethal to many fish at concentrations as low as 0.03 to 0.8 ppm.
-
Swimming Pools and Spas
Some other water-related industries that use chlorine as a disinfectant include swimming pools and spas. It is used to kill microbes such as bacteria, viruses, and algae, and ensure users’ health by preventing the spread of diseases. Chlorine and pH are considered the first line of defense against infection for swimming pools, spas, and hot tubs.
Chlorine is added as a gas, as bleach (sodium hypochlorite), or as other chlorinated products such as calcium hypochlorite, lithium hypochlorite, and chlorinated isocyanurates. A well-managed pool, spa, or tub should, however, not smell of chlorine. Users should avoid swallowing the chlorinated water.
The risk of exposure to the compounds is highest for workers during mixing and applying. The storage and mixing area can be monitored with gas detectors, while protective clothing is required during application.
-
Food Processing Industry
Chlorine is used in the food and beverage industry to address food-borne diseases, a major public health concern.
Cl2 is useful for killing microbes, sanitizing processing equipment and surfaces, pasteurizing cooling water, controlling biofilms, eradicating odors and contamination, and washing fruits and vegetables. Chlorine is also used to wash eggs, meat, fish, poultry carcasses, and equipment surfaces to prevent cross-contamination. Chlorine and its derivatives are used in all stages of food processing. The efficacy of chlorine is influenced by its concentration, contact time, temperature, pH, biofilms, and the presence of organic matter. Chlorine is also used to clean the wastewater from food and beverage processing centers. There are controls to prevent excessive chlorine use, which can harm workers, degrade food sensory quality, and develop malodors.
Chlorine content is critical to the processes, so its concentrations are monitored to maintain process integrity.
-
Wood, Pulp, and Paper
Chlorine gas is used to remove lignin and bleach the pulp, thereby whitening it in kraft paper production. Chlorine reacts with lignin in various processes to produce organochlorine products. During bleaching, chlorine reacts with organic compounds in the wood to produce a range of organochlorine chemicals, many of which are hydrophobic, persistent, and bioaccumulative. Chlorine byproducts enter effluents and are discharged into the environment, posing risks to the food chain and aquatic life. Organochlorines also pose a human health risk.
Safety standards are improving, with pumps and valves controlled remotely rather than manually, reducing chlorine risks. However, accidents can occur. Moreover, over-chlorination of pulp increases the risk of downstream workers’ exposure to chlorine. Workers are also exposed to chlorine during the effluent water treatment processes. Hence, chlorine detectors must be used at the bleaching and water treatment stages when chlorine is added.
-
Textile Industry
Chlorine is also present in the textile industry as a bleaching agent for fabrics. Chlorine is present in many bleaching agents used in fabric production to remove unwanted colors, discoloration, and odor, and for disinfection. One of the main occupational hazards of chlorine in the textile industry is the unpleasant odor, which can cause olfactory function disturbances among workers, 6.33 times greater than those not exposed to chlorine.
Gas monitors are required in storage areas, near feed equipment, processing areas, low-lying areas, and near scrubbing systems.
-
Healthcare Facilities
Chlorine is used to maintain healthcare hygiene and ensure patient safety in hospitals and clinics.
Chlorine is a common sanitizer used to deactivate and kill microbes, including bacteria, viruses, fungi, and spores, in healthcare facilities. Chlorine is used for surface disinfection in many areas of healthcare facilities, including patient wards, operating rooms, intensive care units, and diagnostic areas. It is also used to sterilize equipment and disinfect water to control the spread of diseases and for medical use. Managing residual chlorine in water is essential, and so gas detectors can be used to ensure staff health and safety and process efficiency.
Also, during the COVID-19 pandemic, chlorine derivatives were in high demand for disinfecting surfaces, equipment, and buildings.
-
Electronics and Semiconductor Production
Chlorine is also used in the manufacture of electronics and semiconductors for wet and dry etching and cleaning. For example, the experimental “focused electron beam induced (FEBIE) with chlorine” is used to geometrically shape and modify semiconductor nanodevices. Any residual gas can have a detrimental effect on the itching process, so the gas must be monitored.
-
Oil and Gas
Unconventional oil and gas development (UOGD), namely fracking and horizontal drilling, has increased in the USA. Chlorine gas is released because chlorinated compounds and hydrochloric acid are added to hydraulic fracturing water. Several episodes of high Cl2 gas emissions up to 1 ppb can occur via direct emission. Moreover, chlorine radicals produced in photochemical reactions react with VOCs to form secondary pollutants, such as ozone and organic aerosols, which are harmful to people.
These facilities need gas detectors to ensure that workers are not exposed to Cl2 above permitted levels.
-
Energy Generation
Chlorine chemistry is involved in many alternative and green energy production methods, as well as in conventional power plants.
Seawater is chlorinated and used to cool coal- and nuclear-power stations, transferring excess heat to a heat sink. Water is drawn from the sea and passed into the once-through cooling circuit, which discharges water back into the sea. However, the higher temperatures in the cooling circuit are an ideal habitat for marine life, leading to biofouling. So, chlorine is used as a biocide to kill the marine organisms. The amount of chlorine needed depends on the water’s nutrient levels and organism loads.
Chlorine chemistry is also needed to produce wind turbine blades, solar panels, hybrid car batteries, and ceiling and wall insulation. Chlorine chemistry is also used to produce lightweight components for aircraft and automobiles, thereby improving fuel efficiency.
Areas where chlorine is stored, used, or processed require gas detectors for workers’ safety and health.
Monitoring Chlorine Levels
Monitoring chlorine concentrations to keep them below permitted levels requires reliable, sturdy, and precise technology. Industries will need continuous air monitoring in critical points of their operations where chlorine could be released. Portable tools allow staff to monitor confined spaces for inspections. In addition to air monitoring, ventilation systems to remove Cl2, regular checks, employee training, personal protective equipment, established emergency response plans, and evacuation routes can help reduce risks to personnel and the facility. Industries can consider using Interscan’s fixed gas detectors, AccuSafe, for continuous chlorine monitoring. Interscan’s portable GASD IS for spot checks.
Schedule a demo or meeting to learn more about Interscan’s chlorine detectors.
Sources
Chemical Safety Facts. Org. (2025, June 30). Chlorine in Pools: How Chlorine Keeps Pools Safe. Retrieved from https://www.chemicalsafetyfacts.org/health-and-safety/how-chlorine-keeps-pools-safe-for-summertime-fun/
EMR. (n.d.). Chlorine Market Size, Share and Growth Analysis Report – Forecast Trends and Outlook (2026-2035). Retrieved from https://www.expertmarketresearch.com/reports/chlorine-market
Essential Chemistry Industry.Org. (2018, Nov 27). Chlorine. Retrieved from https://www.essentialchemicalindustry.org/chemicals/chlorine.html
FAO. (n.d.). Use Of Chlorine-Containing Compounds In Food Processing. Retrieved from https://www.fao.org/4/i1357e/i1357e01.pdf
ILO. (2011, March 28). Occupational Hazards and Controls. Retrieved from https://www.iloencyclopaedia.org/part-x-96841/paper-and-pulp-industry/major-sectors-and-processes/item/853-occupational-hazards-and-controls
Masoud, C. G., Modi, M., Bhattacharyya, N., Jahn, L. G., McPherson, K. N., Abue, P., … & Hildebrandt Ruiz, L. (2023). High chlorine concentrations in an unconventional oil and gas development region and impacts on atmospheric chemistry. Environmental Science & Technology, 57(41), 15454-15464.
National Pesticide Information Center. (2024, Jan). Pool and Spa Chemicals Fact Sheet. Retrieved from https://npic.orst.edu/factsheets/pool-chemicals.html
New Jersey Department of Health. (2015, Oct). Hazardous Substance Fact Sheet-Chlorine. Retrieved from https://nj.gov/health/eoh/rtkweb/documents/fs/0367.pdf
Paramerta, N., Nadhifah, D., Marques, M., Saefurrahman, F., Stephanie, I., & Wikaningrum, T. (2024). chlorine residuals in wastewater treatment: implications for pathogen inactivation and environmental impact. Jurnal Penelitian Dan Karya Ilmiah Lembaga Penelitian Universitas Trisakti Учредители: Universitas Trisakti, 341-347.
ScienceDirect. (n.d.). Chlorine (Cl2). Retrieved from
https://www.sciencedirect.com/topics/engineering/chlorine-cl2
Solomon, K. (1996). Chlorine in the Bleaching of Pulp and Paper. Pure and Applied Chemistry, 68(9), 1721-1730. https://doi.org/10.1351/pac199668091721


