- Industrial fires produce more inorganic than organic gases.
- Hazardous gases pose a risk because they can be toxic or asphyxiant, and some, such as carbon monoxide, are lethal at high concentrations in a short time.
- Many gases produced in fires are themselves flammable and will intensify and spread the fire, for example, hydrogen sulfide.
- Other gases are non-flammable but strong oxidizers that can intensify fires, like nitrogen oxides.
Fires are one of the occupational risks that can occur in industries. Several industrial chemicals can form toxic gases during a fire. Inhalation of these toxic gases can increase fatality risks by 86% among firefighters, who are in one of the most dangerous occupations. Identifying the toxic gases formed in fire and the associated risks is challenging. This article includes some of the most common toxic gases that can form in fires and the sources from which they arise, which Safety managers should be aware of to alert firefighters in the event of a fire at their facilities.
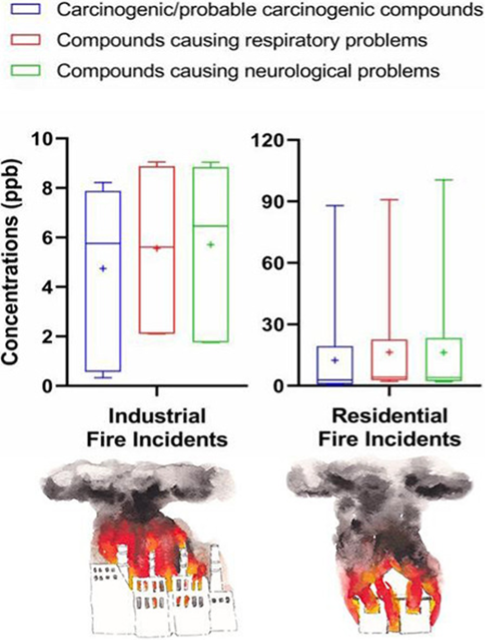
Figure 1. A study comparing various fire accidents found that gases leading to cardiovascular and neurological diseases and cancers were higher in industrial fires Alharbi et al., (2021). (Image credits: DOI 10.1016/j.scitotenv.2021.145332)
Gas Formation in Industrial Fires
Besides industries that use chemicals, waste and recycling facilities are also sites of accidental fires that produce significant amounts of toxic gases, involving waste materials such as tires, paper, wood, cardboard, plastics, timber, electrical and electronic waste, and residual mixed waste.
Any fire will produce smoke, vapors, and gases. Each poses different risks to occupants or to firefighters entering to contain the fire:
- Smoke: In any fire, the danger is most from smoke. It is the most visible product of combustion and is composed of particulate matter. The danger from smoke is reduced visibility, which can hinder escape and firefighting efforts.
- Vapor: Formed as a gas due to combustion and high temperature, vapors can revert to their solid or liquid forms at normal temperature. Vapors can be toxic at sufficiently high concentrations.
- Gases: Toxic gases are products of chemical reactions that occur during a fire and remain gases even at normal temperatures. Gases can be toxic when inhaled or absorbed through the skin and eyes.
Toxic gases and vapors can be asphyxiants, respiratory irritants, and toxicants with other effects. The effects depend on the concentrations of the gases and the duration of exposure. Even small amounts can cause irritation of the skin, eyes, and respiratory tract. Many gases formed during a fire are combustible and can prolong and spread the fire. Toxic gases and vapors can lead to fatalities immediately or due to chronic, irreversible illnesses like cardiovascular and neurological diseases and cancers; see Figure 1. Therefore, personal protective equipment with a self-breathing apparatus is essential for firefighters.
Besides toxicity, the gases produced in fires can also be flammable or oxidizers.
Fire Chemistry
The gases formed during a fire are determined by the fire chemistry.
Fire needs an ignition source, fuel or material that burns, and an oxidizer. The oxidizer is usually oxygen (O2) in the air. However, many other chemicals can also act as oxidizers, such as chlorine, bromine, fluorine, nitrates, nitrites, bromates, peroxides, chlorates, and persulphates. The products formed by these oxidizers can differ from those formed by fires involving oxygen. Several oxidizers cannot be controlled by water; in fact, they react with water to form oxygen or other gases that will intensify fires.
Challenges in Gas Identification
Thus, the hazardous gases produced depend on the fire chemistry: fuel type, stage of fire, combustion conditions, composition, and amount of burned materials, and are therefore difficult to predict exactly. For example, methane, gasoline, and fuel oils produce 2.5 times the heat as wood and paper.
The invisible and odorless nature of most toxic gases also makes their detection and identification challenging.
However, depending on the chemical composition of materials involved in a fire, it is possible to narrow down the gases that may be present, helping industries and firefighters reduce losses.
Most fires involve wood, polymers like plastic and rubber, and textiles. It is also pertinent to remember that many building materials also contain chemicals, such as polymers, that burn and emit gases. In industrial fires, firefighters are at a greater risk from inorganic gases, according to a study by Alharbi et al. (2021).
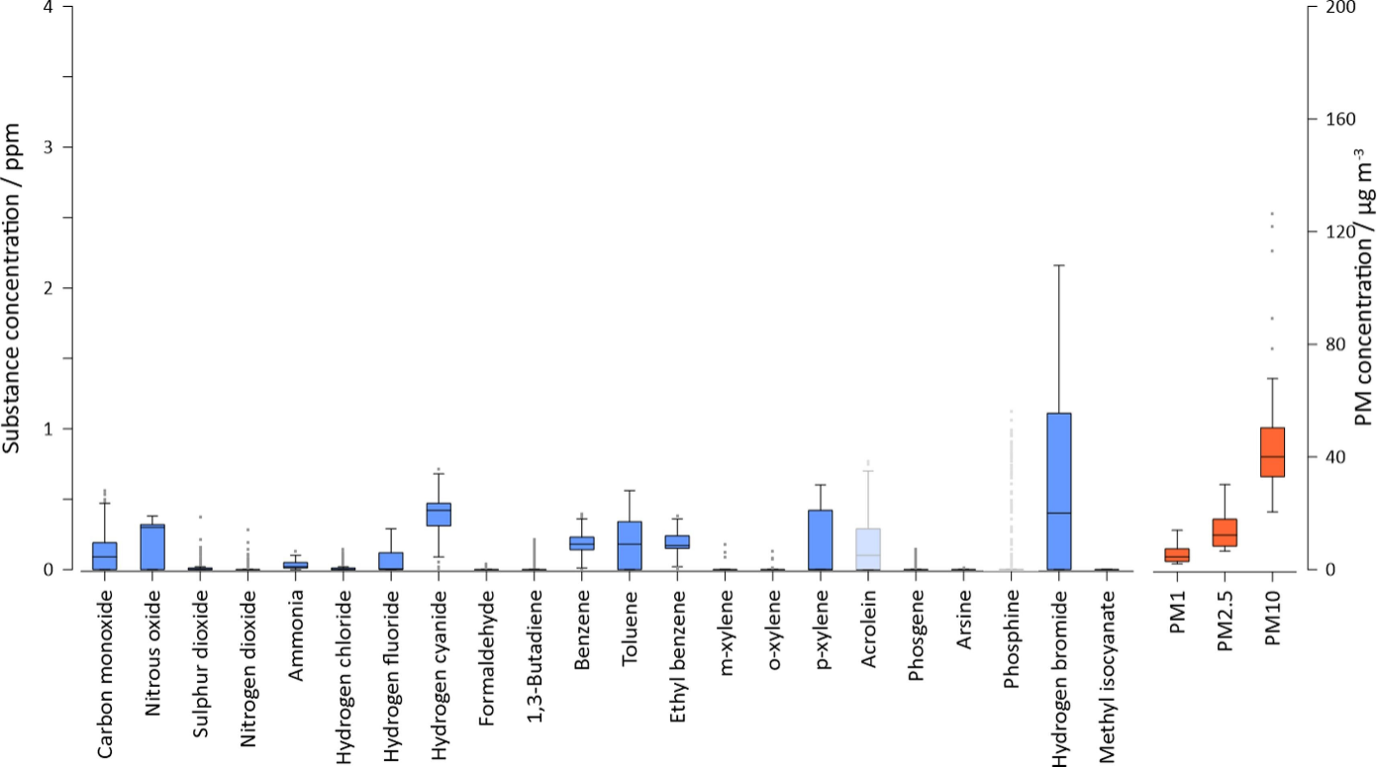
Figure 2: “Substances measured during a fire at a chemical plant manufacturing pesticides.‘Ghosted’ boxes indicate substances where significant cross-interference from other substances is likely, and so should be considered as less reliable,” Griffiths et al. (2018). (Image credits: https://doi.org/10.1016/j.envint.2022.107152)
The hazardous inorganic and organic gases produced in industrial fires are discussed below; also see Figure 2.
-
Carbon Monoxide
Carbon monoxide (CO) is a colorless and odorless gas, which is produced in significantly higher quantities than any other hazardous gas during a fire. It is toxic and flammable.
CO forms during flaming and smoldering fires. Carbon monoxide is formed by the incomplete combustion of carbon-containing materials in the presence of oxygen, such as wood, paper, fossil fuels, and organic polymers. CO is the main product of polymer combustion. Several organic polymers, made of carbon and hydrogen, are used to make consumer products and building materials, and these chemicals are widespread, such as polyethylene, polystyrene, and acrylic fibers. Plexiglass, made of methylmethacrylate, breaks down to smaller compounds that react with O2 to form CO.
CO is dangerous to people because it is an asphyxiant. CO has a greater affinity for hemoglobin in the red blood cells than O2, and forms irreversible carboxyhemoglobin. CO reduces the blood’s capacity to transport O2, which can affect several organs and be fatal in high doses. In smaller quantities, it causes headaches, dizziness, and fatigue. CO is responsible for many deaths that involve wood and cellulose.
More details on CO effects and safe limits are available in the New Jersey Department of Health Hazardous Substance Fact Sheet.
-
Carbon Dioxide
Carbon dioxide (CO2) is also formed when any substance containing carbon burns, and it is produced by most fires, especially smoldering fires. The gas is colorless and odorless, asphyxiant and poisonous, but non-flammable.
The amount of CO2 in the air will reduce O2 levels, making it an asphyxiant. Also, inhaling CO2 increases breathing, so there is a risk that people will inhale more air, laden with toxic gases. This toxic effect starts when CO2 is present above 5% gas concentration or 50,000 ppm, and intoxication effects appear after 30 minutes of exposure. The safe limits for a day are 5000 ppm for people.
More details on CO2 effects and safe limits are available in the New Jersey Department of Health Hazardous Substance Fact Sheet.
-
Hydrogen Cyanide
Hydrogen cyanide (HCN) is a colorless gas above 78 °F (26 °C); below this temperature, it is a blue liquid. It has a faint, bitter-almond odor, is toxic, and is highly flammable.
HCN is produced when materials containing nitrogen burn. These can be wool, silk, urea-formaldehyde, and polymers such as nylon, orlon (polyacrylonitrile), polyurethane, acrylic carpeting materials, and ABS (acrylonitrile-butadiene-styrene) pipes. HCN is the main product of burning wool. More HCN is produced when plastics burn than when wood and fossil fuels burn. HCN formation also depends on temperature.
HCN is toxic because it inhibits the function of body tissues.
More details on hydrogen cyanide effects and safe limits are available in the New Jersey Department of Health Hazardous Substance Fact Sheet.
-
Hydrogen Chloride
Hydrogen chloride (HCl) is a colorless to slightly yellowish gas with a pungent odor that fumes in the air. It is highly corrosive and toxic but non-flammable.
HCl is formed when chlorine-containing materials are burnt, such as chlorinated plastics like polyvinyl chloride (PVC), a widely used plastic. It is used to make industrial cables and pipes. Over 40% of the compounds formed when PVC burns are HCl, and HCl formation can exceed that of CO. HCL content is 18 times higher in industrial fires than in residential fires.
It is not absorbed through the skin and is harmful only when inhaled. However, HCl gas can react with water to form hydrochloric acid that can irritate and burn the skin. At high concentrations, it is corrosive to the skin, eyes, mucous membrane, nose, and respiratory and gastrointestinal tracts. HCl harms the upper respiratory tract and is immediately dangerous to life or health (IDLH) at 50 ppm.
More details on hydrogen chloride effects and safe limits are available in the New Jersey Department of Health Hazardous Substance Fact Sheet.
-
Hydrogen Bromide
Hydrogen bromide (HBr) is colorless and has a strong and irritating odor. It is toxic and corrosive but non-flammable. When HBr reacts with water, it can form hydrobromic acid.
HBr is produced when materials containing bromine flame-retardant burn. Polymers used in industry often contain bromine flame retardants, for example, in printed circuit boards, electronics, computers, and cell phones.
HBr is toxic on inhalation, and its corrosivity can affect the skin, eyes, nose, and throat.
More information on hydrogen bromide is available in the New Jersey Department of Health Hazardous Substance Fact Sheet.
-
Hydrogen Fluoride
Hydrogen Fluoride (HF) is a fuming, colorless gas with a strong, irritating odor. It is toxic and corrosive, but non-flammable. It dissolves in water to form hydrofluoric acid.
HF is formed when materials containing fluorine burn, such as several inorganic compounds, fluorite (used in metallurgy), lithium-ion batteries, and fluoropolymers (high-performance plastics) used in many industries, including renewable energy, advanced electronics, healthcare, semiconductors, pharmaceuticals, and food production
Breathing HF at low levels can irritate the eyes, nose, throat, and lungs. At higher concentrations, it causes death due to irregular heartbeat or fluid buildup in the lungs. HF is Immediately Dangerous to Life or Health (IDLH) at 30 ppm.
More information on hydrogen fluoride is available in the New Jersey Department of Health Hazardous Substance Fact Sheet.
-
Hydrogen Sulfide
Hydrogen sulfide (H2S) is a colorless gas with the smell of rotten eggs at lower concentrations below 100 ppm. At concentrations above these levels, H2S is not detectable because it causes smell paralysis. H2S is highly toxic, corrosive, and flammable.
H2S is formed due to the burning of material containing sulfur, such as wool, leather, vulcanized rubber, meat, and hair. Hydrogen sulfide production in industrial fires is 19 times higher than in residential fires.
H2S affects the respiratory and neurological systems, causing headaches, weakness, dizziness, nausea, shortness of breath, palpitations, rapid respiratory rate, bronchitis, pulmonary edema, and respiratory paralysis. According to the National Institute for Occupational Safety and Health (NIOSH), IDHL is at 100 ppm.
More information on hydrogen sulfide is available in the New Jersey Department of Health Hazardous Substance Fact Sheet.
-
Sulfur Dioxide
Sulfur dioxide (SO2) is a colorless gas with a pungent, irritating smell. It is toxic and non-flammable.
SO2 is formed when sulfur-containing materials burn, like polymers (plastics and rubber), fossil fuels, wool, wood, leather, and other organic matter. SO2 is produced in smaller quantities, but its production is 8 times higher in industrial fires than in residential fires.
SO2 affects humans through contact and inhalation. It can irritate the skin, eyes, nose, and throat, and at higher concentrations, it causes pulmonary edema.
More information on sulfur dioxide effects and safe limits is available in the New Jersey Department of Health Hazardous Substance Fact Sheet.
-
Phosgene
Phosgene (COCl2) is a colorless to slightly yellow colored gas with a suffocating, newly mown hay smell. The odor threshold is five times the permitted limit, so smell is not useful for detection. The gas is toxic and corrosive. It can interact with water to slowly form hydrochloric acid.
Phosgene is generated by the combustion of any chlorine-containing materials, especially in confined spaces. Sources of phosgene include polymers such as polyvinyl chloride (PVC), solvents, dry-cleaning agents, metal parts, and refrigerants.
Phosgene is only inhaled and not absorbed through the skin. However, phosgene reacts with water, so contact with moist surfaces, such as the eyes or mucous membranes, can cause irritation of the affected organs. The gas is heavier than air and sinks to the floor, and can cause asphyxiation in poorly ventilated spaces. Higher levels can cause pulmonary edema.
More information on phosgene effects and safe limits is available in the New Jersey Department of Health Hazardous Substance Fact Sheet.
-
Nitrogen Oxides
Several nitrogen oxides (NOx) are formed in industrial fires, like nitrous oxide (N2O), nitric oxide (NO), nitrogen dioxide (NO2), and nitrogen tetroxide (N2O4). They react with moisture in the respiratory tracts to produce nitrous and nitric acids.
The nitrogen oxides are formed when materials containing nitrogen, like protein-rich wool and silk, fossil fuels (propane, diesel, and gasoline), wood, and paper, are burned.
Nitrous oxide: N2O is colorless with a sweet odor and taste. It can irritate the eyes, nose, and throat, causing a pins-and-needles sensation in the limbs, and higher acute exposures can cause death.
Nitric oxide: Nitric oxide, or nitrogen monoxide, is quickly converted to NO2 in air. It is colorless and has a sharp odor. It is toxic, a strong oxidizer, and a fire and explosion risk. It affects the eyes, skin, and the respiratory system. High levels of NO reduce the blood’s ability to transport O2, causing blue skin and lips (methemoglobinemia).
Nitrogen dioxide: NO2 is a brownish-red gas with a strong odor. It is very toxic and a strong oxidizer that can cause explosions. NO2 is as toxic as hydrogen cyanide, and it can lead to serious respiratory problems and methemoglobinemia.
Nitrogen tetroxide: Also called dinitrogen tetroxide, it is a brown vapor. It is produced by the combustion of ammonium nitrate, nitrocellulose, and organic nitrates. It is a strong oxidizer that can intensify fires. It causes skin burns, eye damage, choking, headache, and chest pain. Other symptoms develop after 5-72 hours, like difficulty in breathing, methemoglobinemia, pulmonary edema, and death by asphyxiation.
-
Methane
Methane (CH4) is a colorless and odorless gas that is toxic and highly flammable.
Methane is produced by the incomplete combustion of fossil fuels in industrial processes, and by accidental fires in waste recycling facilities and mines.
Methane can lower O2 levels at high concentrations and act as an asphyxiant, leading to headache, weakness, loss of judgment and coordination, and unconsciousness, which can hamper firefighting activities. Being flammable, it can intensify and spread the fire.
More information on methane is available in the New Jersey Department of Health Hazardous Substance Fact Sheet.
-
Volatile Organic Compounds
Exposure to volatile organic compounds (VOCs) among firefighters is 50% lower in industrial fires than in residential fires.
Many VOCs are formed in industrial fires. These can result from burning polymers including synthetic rubber, wood, cellulose, hemicellulose, lignin, textiles, and natural rubber.
The VOCs usually formed by fires are 1,3-butadiene, benzene, acrolein, ethylbenzene, toluene, xylenes, styrene, formaldehyde, arsine, methyl isocyanate, etc. Several other VOCs are also produced to a lesser extent. Several VOCs, like toluene, benzene, ethylbenzene, xylenes, acrolein, and styrene, are formed by the incomplete combustion of materials.
VOCs, like formaldehyde, are toxic and carcinogenic. VOCs make up a significant portion of aerosols found in fires and contribute to their flammability.
Air Monitoring
Firefighters use portable multigas detectors when fighting fires. Industries can monitor air quality in their facilities using fixed monitors equipped with multiple gas sensors for various gases, especially in industries that involve fuel combustion. Several operations, such as metal welding, that routinely produce CO, hydrogen fluoride, phosgene, and nitrogen oxides can have the appropriate gas detectors to alert workers to these levels. Interscan provides fixed gas detectors for multiple gases and portable devices for specific gases. Interscan detectors cover most of the gases produced by fires and can also be used by scientists to study hazardous gases produced as fire byproducts.
Contact us for more information on Interscan gas detectors.
Sources
Alharbi, B. H., Pasha, M. J., & Al-Shamsi, M. A. S. (2021). Firefighter exposures to organic and inorganic gas emissions in emergency residential and industrial fires. Science of the Total Environment, 770, 145332. DOI 10.1016/j.scitotenv.2021.145332
Austin, C. (1997). Municipal firefighter exposures to toxic gases and vapours. [Doctoral dissertation, McGill University, Canada]. Retrieved from https://www.researchgate.net/profile/Claire-Austin/publication/30007794_Firefighter_exposure_to_toxic_gases_and_vapours/links/544d59e20cf2bcc9b1d8ea14/Firefighter-exposure-to-toxic-gases-and-vapours.pdf
Barontini, F., Marsanich, K. & Cozzani, V. (2004). The use of TG-FTIR technique for the assessment of hydrogen bromide emissions in the combustion of BFRs. Journal of Thermal Analysis and Calorimetry 78, 599–619. https://doi.org/10.1023/B:JTAN.0000046122.00243.ed
Burgess, H. (2025, Dec, 1). Oxidizers Pose Unique Fire and Explosion Risks. Retrieved from https://www.nfpa.org/news-blogs-and-articles/blogs/2024/10/11/georgia-chemical-plant-fire-and-oxidizers
Gann, R. & Bryner, N. P. (n.d.). Section 6, Chapter 2 Combustion Products and Their Effects on Life Safety. In Characteristics of Materials and Products. (pp-11-34). Retrieved from https://tsapps.nist.gov/publication/get_pdf.cfm?pub_id=900093
Griffiths, S. D., Chappell, P., Entwistle, J. A., Kelly, F. J., & Deary, M. E. (2018). A study of particulate emissions during 23 major industrial fires: Implications for human health. Environment International, 112, 310-323.https://doi.org/10.1016/j.envint.2022.107152
Sumi, K., & Tsuchiya, Y. (1971). Toxic gases and vapors produced at fires. Retrieved from DOI: https://doi.org/10.4224/40000748
Tupper C, Doyal A. [Updated 2023 Feb 27]. OSHA Fire Safety. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK580481


